Availability of narcotic drugs for medical use
The Board has an important role to play in the supply of raw materials required for the manufacture of all medications containing opiates. At the same time the Board strives, in cooperation with Governments, to maintain a lasting balance between supply of and demand for those materials. The analysis of the data provided by Member States shows that the amount of opiate raw material available for the manufacturing of narcotic drugs for pain relief is more than sufficient to satisfy the current level of demand as reported by Governments and that global stocks are increasing.
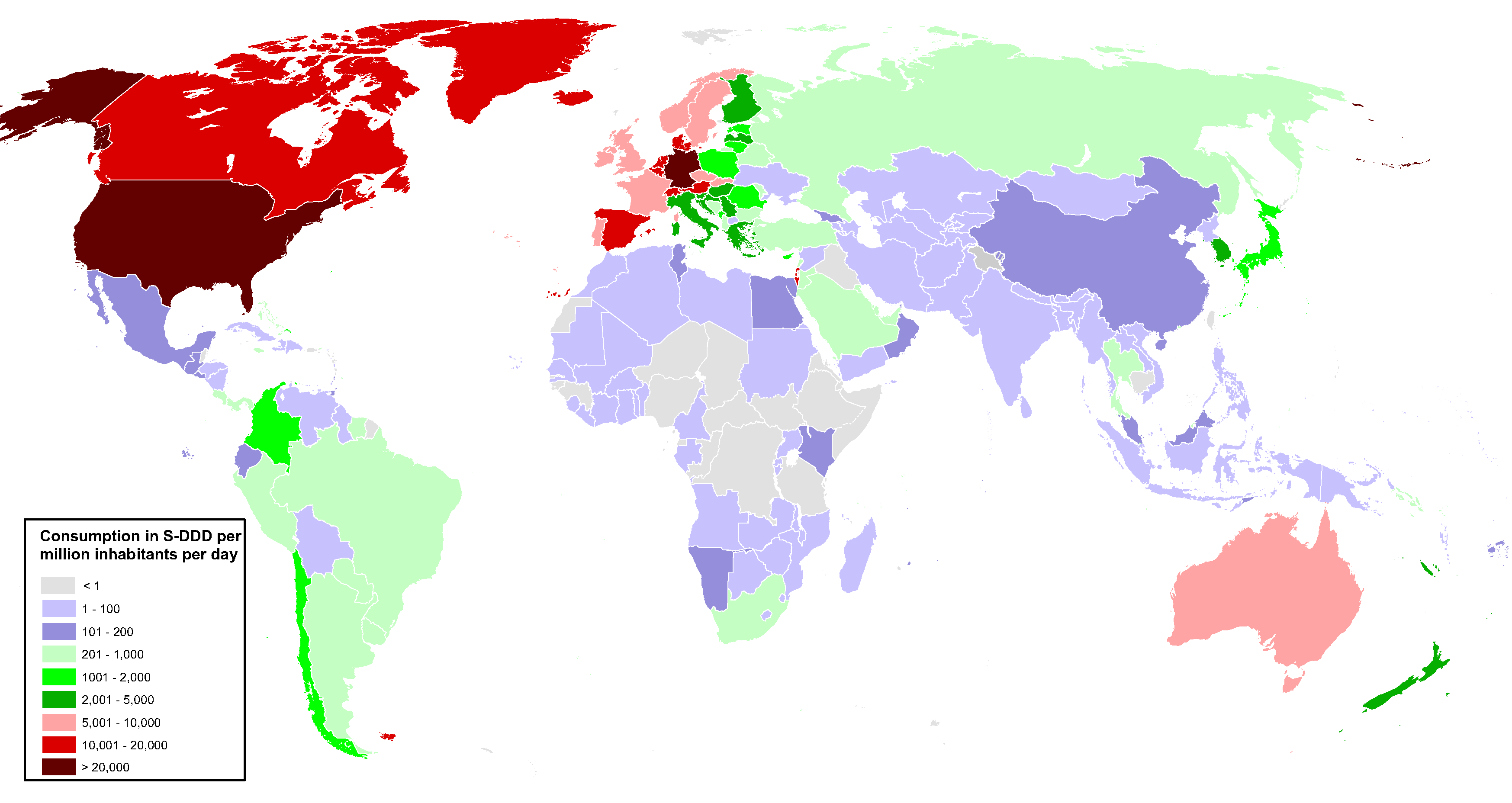
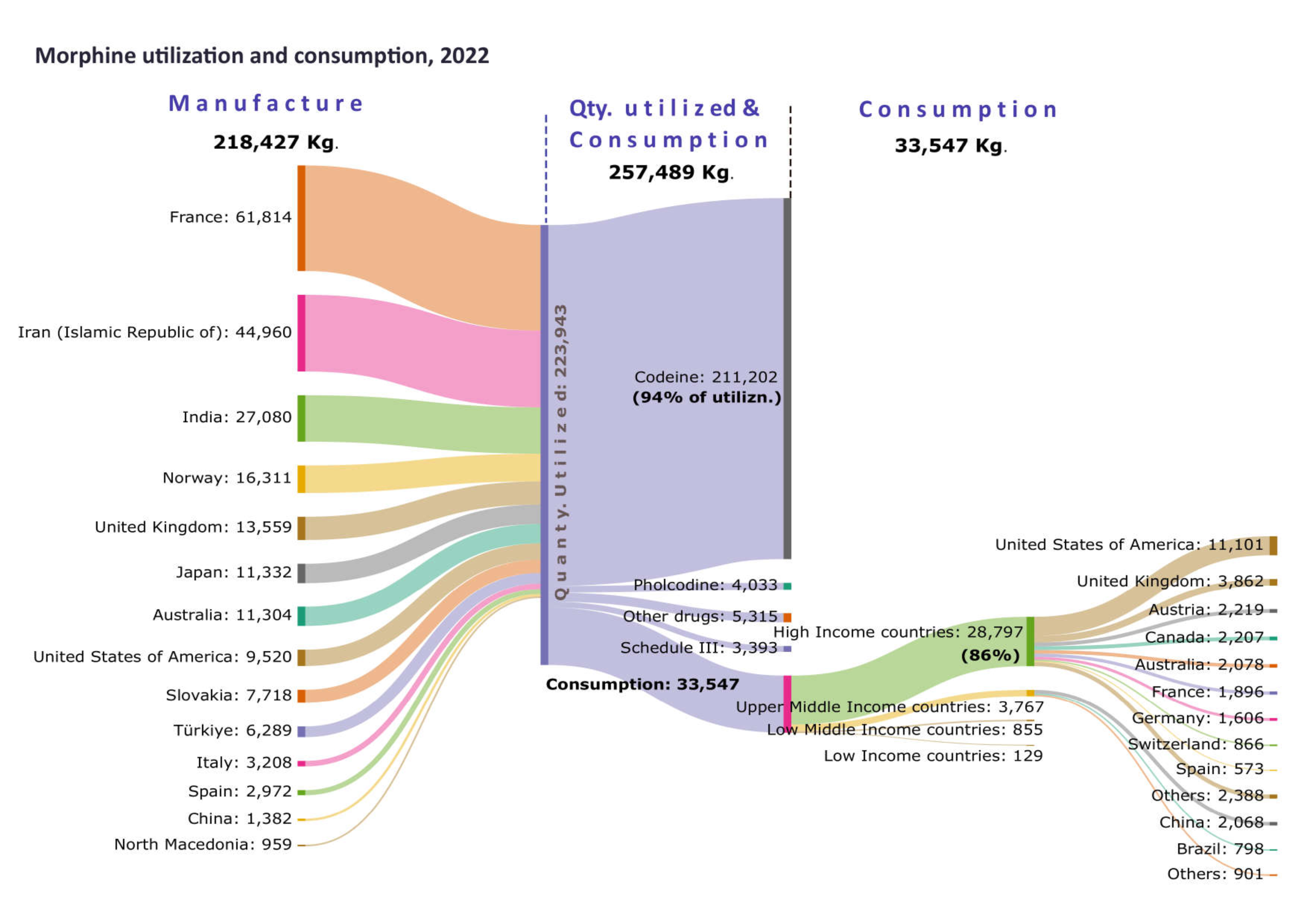
The majority of morphine (94 per cent, on average) was converted into codeine or into substances not covered by the 1961 Convention. Most of the codeine manufactured (89 per cent) was used to manufacture cough medication. In 2022, of the limited amount (less than 10 per cent) of morphine used directly for pain management, a small percentage (14 per cent) was available in countries constituting 80 per cent of the world population. The remaining 86 per cent of available morphine, excluding preparations included in Schedule III of the 1961 Convention, was concentrated in a small number of countries located mainly in Europe and North America.
|
This imbalance is particularly problematic because latest data available show that over 70 per cent of cancer deaths actually occur in low- and middle-income countries. Without sustained action, cancer incidence is projected to increase by 70 per cent in middle-income countries and 82 per cent in lower-income countries by 2030.
Apart from the needs related to cancer, pain treatment is needed in many other medical situations (surgery, delivery, etc.) and in several regions pain relief drugs are not commonly prescribed. Other internationally controlled drugs such as methadone and buprenorphine (an opioid analgesic which is controlled under the 1971 Convention and whose use in substitution therapy continues to increase) are used in the management of drug dependence and their use is also limited in some countries despite considerable prevalence of heroin abuse.
As long as these drugs remain inaccessible to the large majority of people around the world, patients will not be able to derive the health benefits to which they are entitled under the Universal Declaration of Human Rights. In other countries, however, overprescribing opioid analgesics and their availability in quantities greater than those required for sound medical treatment may lead to the diversion and abuse of those substances with negative consequences such as overdose and addiction.
In 2010, INCB published a report on the availability of internationally controlled drugs in which the Board highlighted the impediments to the availability of opioids analgesics as reported by countries. These impediments include regulatory, attitudinal, knowledge-related, economic and procurement-related factors that can adversely affect availability. In 2010 the most important impediments listed by countries were concerns about addiction, reluctance to prescribe or stock and insufficient training for professionals. Unduly restrictive laws and burdensome regulations were also commonly perceived as playing a significant role in limiting the availability of opioids. A smaller number of Governments reported that difficulties involving distribution and supply and the high cost of opioids were major obstacles to making opioids adequately available.
In 2018, INCB published a report Progress in ensuring adequate access to internationally controlled substances for medical and scientific purposes as a review of the implementation of recommendations, on the availability and access to controlled substances, contained in its 2010 report.
In 2022, INCB published a report Supplement to the annual report of the Board for 2022 on the availability of Internationally Controlled Substances: No Patient Left Behind: Progress in Ensuring Adequate Access to Internationally Controlled Substances for Medical and Scientific Purposes as an update on the availability and access to controlled substances, contained in its 2018 report.
INCB urges countries and governments to implement the recommendations contained in the 2022 report with the overall goal of ensuring a well-functioning national and international system for managing the availability of narcotic drugs and psychotropic substances that should provide relief from pain and suffering by ensuring the safe delivery of the best affordable drugs to those patients who need them and, at the same time, prevent the diversion of drugs for the purpose of abuse.
|